

新疆农业科学 ›› 2022, Vol. 59 ›› Issue (4): 990-1000.DOI: 10.6048/j.issn.1001-4330.2022.04.025
• 基因工程·草业·农产品分析检测·畜牧兽医 • 上一篇 下一篇
收稿日期:2021-03-24
出版日期:2022-04-20
发布日期:2022-04-24
通信作者:
王艳
作者简介:地力努尔·艾力木江(1997- ),女,新疆人,硕士研究生,研究方向为植物学,(E-mail) 1455636632@qq.com
基金资助:
Delinur Alimujiang( ), FENG Xiaoli, WANG Yan(
), FENG Xiaoli, WANG Yan( )
)
Received:2021-03-24
Online:2022-04-20
Published:2022-04-24
Correspondence author:
WANG Yan
Supported by:摘要:
【目的】研究盐生植物盐穗木HcSCL13基因的时空表达和胁迫响应特性。【方法】将该基因全长启动子(2 230 bp)及截短序列与GUS报告基因融合,检测遗传转化后植物的GUS组织化学染色及酶活力。【结果】在稳定整合HcSCL13启动子的转基因拟南芥中,从种子萌发到种苗形成的过程中,地上和地下部位均表现出较强的启动子活性;随着植物的生长发育,启动子活性表现出一定的组织特异性;HcSCL13基因启动子对光、盐及机械损伤等胁迫积极响应。启动子上游-1 124-1 450 bp是影响其活性的区域,而上游-1 730-2 230 bp为盐响应区域。【结论】盐穗木HcSCL13基因的表达具有时空特异性及对光、盐和机械损伤等因素的响应特性。
中图分类号:
地力努尔·艾力木江, 冯肖莉, 王艳. 盐穗木转录因子HcSCL13基因的时空表达和胁迫响应特性[J]. 新疆农业科学, 2022, 59(4): 990-1000.
Delinur Alimujiang, FENG Xiaoli, WANG Yan. Study of the Promoter properties of the HcSCL13 Gene for the Transcription Factor of Halostachys caspica (Bieb.) C. A. Mey.[J]. Xinjiang Agricultural Sciences, 2022, 59(4): 990-1000.
| 名称 Name | 序列(5'- 3') Sequence |
|---|---|
| PHcSCL13-SP1P1 | GATTACGCCAAGCTTAATTAGATCT GTTTGCAACC |
| PHcSCL13-SP2 P1 | GATTACGCCAAGCTTTCCCAATAGC CTCATATCTC |
| PHcSCL13-SP3 P1 | GATTACGCCAAGCTTCAA AACCAGTCTA ATAGCCG |
| PHcSCL13-SP4 P1 | GATTACGCCAAGCTTTGAATTGACC AGCTTGATTG |
| PHcSCL13-SP5 P1 | GATTACGCCAAGCTTTTCAACTATT TCCGTACAATTT |
| PHcSCL13P2 | CCGGGGATCCTCTAGACCGAGAATAG CACAATCTGA |
表1 HcSCL13 截短启动子的引物设计
Table 1 Primer design of HcSCL13 truncated promoter
| 名称 Name | 序列(5'- 3') Sequence |
|---|---|
| PHcSCL13-SP1P1 | GATTACGCCAAGCTTAATTAGATCT GTTTGCAACC |
| PHcSCL13-SP2 P1 | GATTACGCCAAGCTTTCCCAATAGC CTCATATCTC |
| PHcSCL13-SP3 P1 | GATTACGCCAAGCTTCAA AACCAGTCTA ATAGCCG |
| PHcSCL13-SP4 P1 | GATTACGCCAAGCTTTGAATTGACC AGCTTGATTG |
| PHcSCL13-SP5 P1 | GATTACGCCAAGCTTTTCAACTATT TCCGTACAATTT |
| PHcSCL13P2 | CCGGGGATCCTCTAGACCGAGAATAG CACAATCTGA |
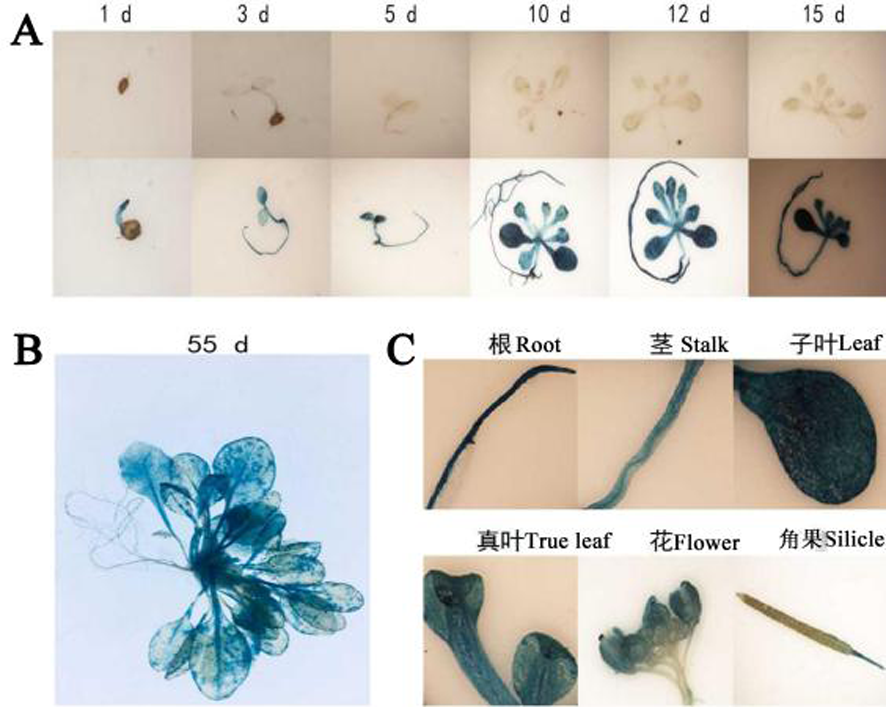
图2 HcSCL13 启动子的时空表达模式 注:A:种苗阶段,B:成苗阶段,C:生殖阶段的不同组织部位
Fig. 2 Spatial-temporal expression patterns of HcSCL13 promoter Note:A:Seedling stage, B : Adult seedling, C : Different tissues at the reproductive stage
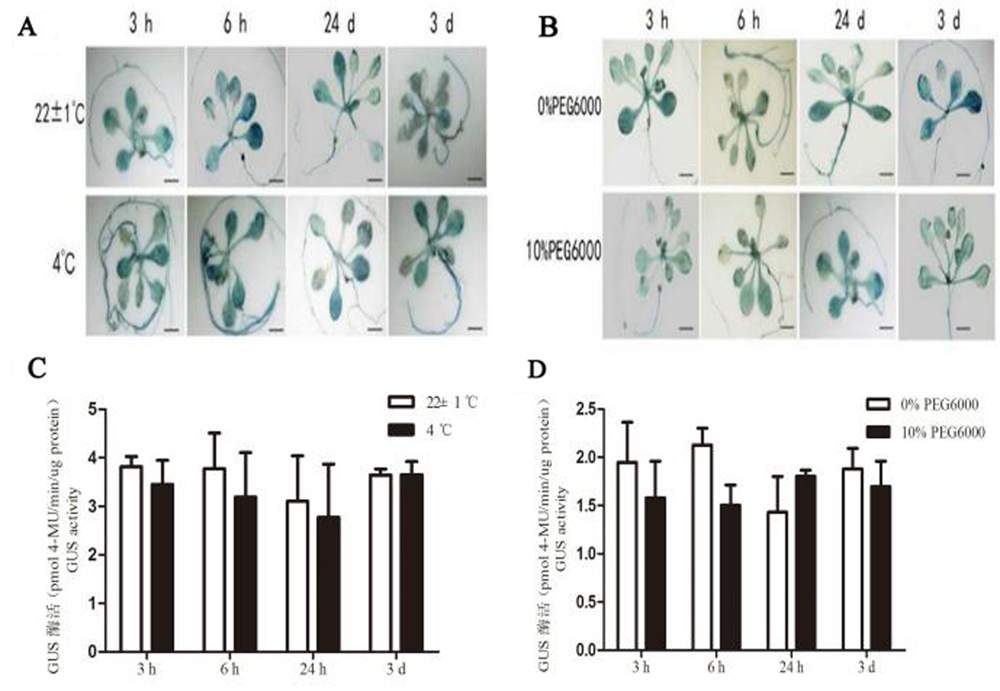
图5 HcSCL13 启动子对低温和干旱胁迫的响应 注:A 低温胁迫下的GUS组织化学染色;B干旱胁迫下的GUS组织化学染色;C 低温胁迫下的GUS酶活;D干旱胁迫下的GUS酶活
Fig. 5 Response to low temperature and drought stress for HcSCL13 gene promoter Note: A:GUS staining under low temperature, B: GUS staining under drought condition, C: Activity analysis for GUS, D Activity analysis for GUSunder drought condition
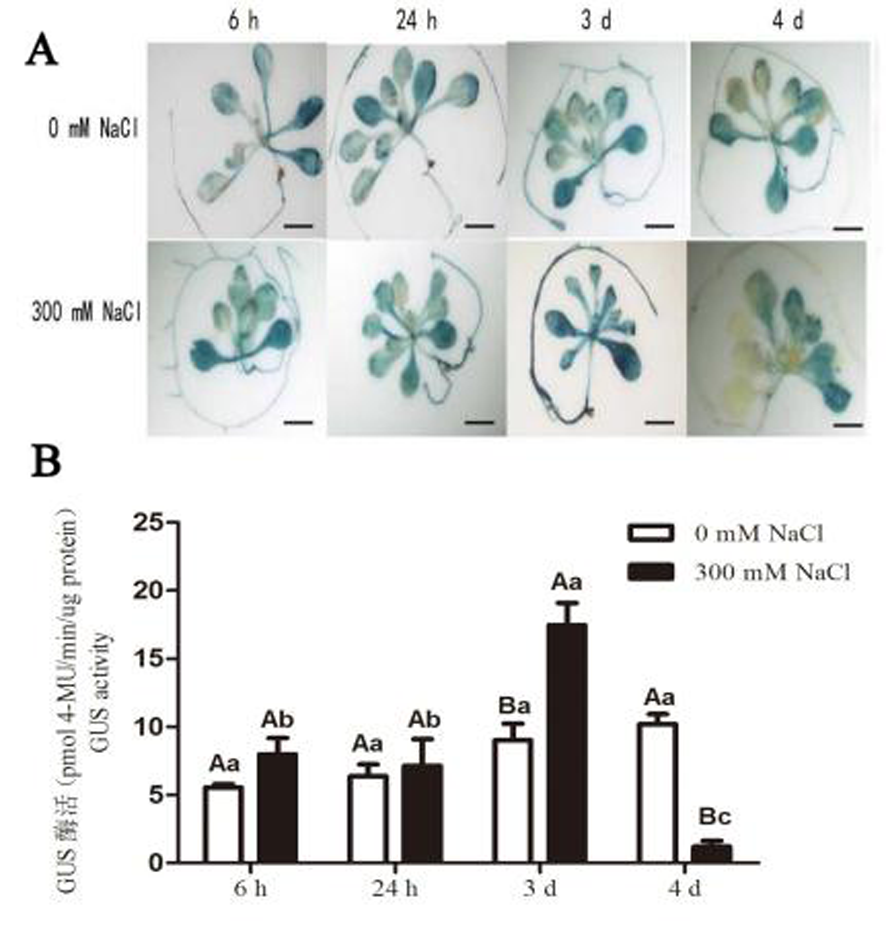
图6 HcSCL13启动子下盐胁迫的响应变化 注:A:GUS组织化学染色,B:GUS酶活;数值为3次平行实验的平均值±标准差,不同小写字母代表组间差异(P< 0.05),不同大写字母代表组内差异(P< 0.05)
Fig. 6 Response to salt stress for HcSCL13 gene promoter Note: A:GUS staining,B:Activity analysis for GUS; The value is the mean ± standard deviation of three parallel experiments. Different lowercase letters represent the difference between groups (P< 0.05), and different uppercase letters represent the difference within groups (P< 0.05)
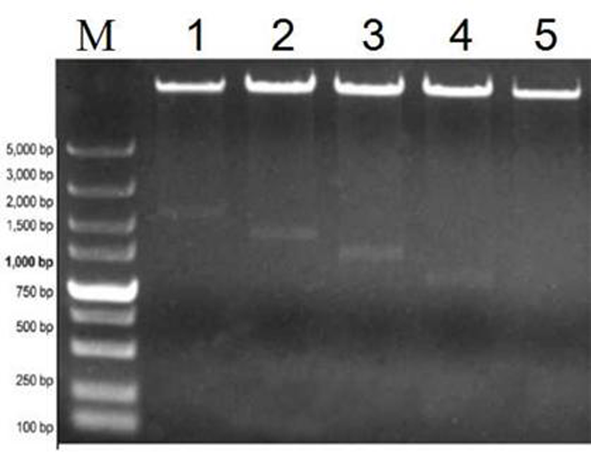
图7 HcSCL13截短启动子植物表达载体的酶切鉴定 注:M:5 000 bp Marker,1-5:HindⅢ和XbaⅠ分别双酶切重组质粒pBI121-PHcSCL13SP1, pBI121-PHcSCL13SP2, pBI121- PHcSCL13 SP3, pBI121-PHcSCL13SP4和pBI121-PHcSCL13SP5
Fig. 7 Identification of plant expression vector of truncated HcSCL13 promoter Note: M:5,000 bp marker; 1-5: Recombinant plasmids of pBI121-PHcSCL13SP1,pBI121-PHcSCL13SP2,pBI121 -PHcS CL13SP3, pBI121-PHcSCL13SP4 and pBI121-PHcSCL13SP5 were digested by Hind Ⅲ and Xba I, respectively
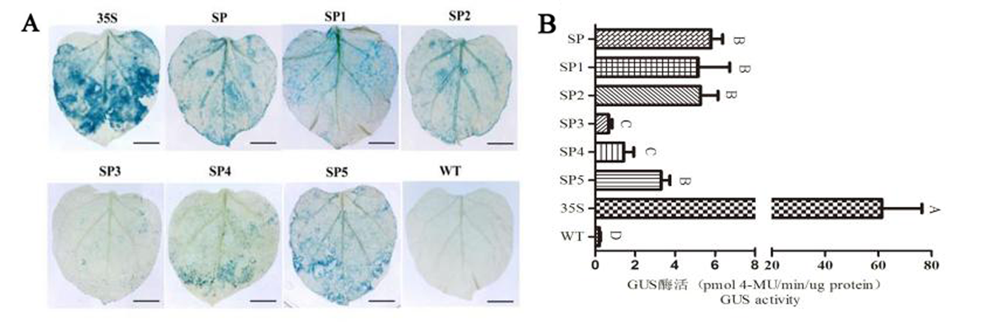
图8 HcSCL13截短启动子在瞬转染烟草中的活性 注:A:GUS组织化学染色,B:酶活分析;数值为4次平行实验的平均值±标准差,不同大写字母代表组间差异(P< 0.05)
Fig.8 Analysis oftruncated HcSCL13 promoterin transiently transfected tobacco Note: A:GUS staining, B:Activity analysis for GUS;The value is the mean ± standard deviation of 4 parallel experiments, and different capital letters represent the difference between groups (P< 0.05)
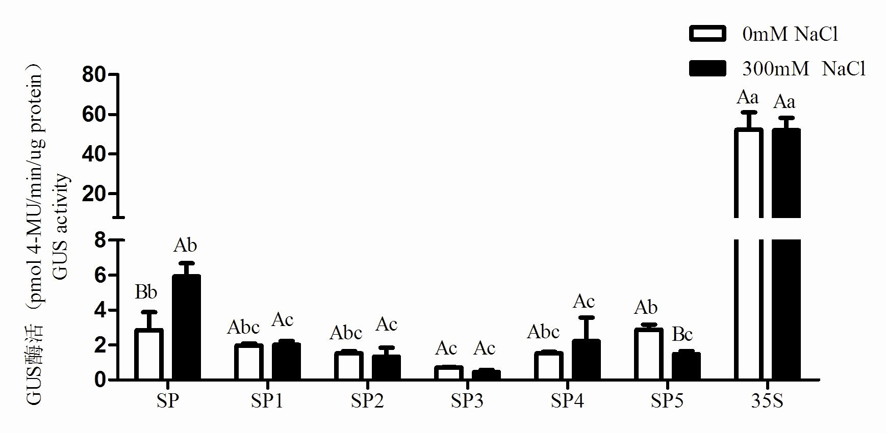
图9 盐胁迫下截短启动子活性 注:数值为4次平行实验的平均值±标准差,不同小写字母代表组间差异(P< 0.05),不同大写字母代表组内差异(P< 0.05)
Fig. 9 Analysis of truncated promoter activity under salt stress Note: The value is the mean ± standard deviation of four parallel experiments. Different lowercase letters represent the difference between groups (P< 0.05), and different uppercase letters represent the difference within groups (P< 0.05)
| [1] | 陈忠良, 李圣彦, 汪海, 等. 玉米转录因子EREB58启动子分析[J]. 中国农业科技导报, 2016, 18(6):23-30. |
| CHEN Zhongliang, LI Shengyan, WANG Hai, et al. Promoter analysis of maize transcription factor EREB58[J]. China Agricultural Science and Technology Guide, 2016, 18(6):23-30. | |
| [2] | 张亚洲, 陈芬, 徐庆国, 等. 转录因子DREB在植物抗逆中的应用[J]. 作物研究, 2009, 23(5):290-292. |
| ZHANG Yazhou, CHEN Fen, XU Qingguo, et al. Application of transcription factor DREB in plant stress resistance[J]. Crop Research, 2009, 23(5):290-292. | |
| [3] | 王金英, 丁峰, 潘介春, 等. 植物bZIP转录因子家族的研究进展[J]. 热带农业科学, 2019, 39(6):39-45. |
| WANG Jinying, DING Feng, PAN Jiechun, et al. Research progress of plant bZIP transcription factor family[J]. Tropical Agricultural Science, 2019, 39(6):39-45. | |
| [4] |
Sun X L, Xue B. A functionally required unfoldome from the plant kingdom: intrinsically disordered N-terminal domains of GRAS proteins are involved in molecular recognition during plant development[J]. Plant Molecular Biology, 2011, 77(3):205-223.
DOI URL |
| [5] |
Chumacher, Karin, Schmitt, et al. The Lateral suppressor (Ls) gene of tomato encodes a new member of the VHIID protein family[J]. Proceedings of the National Academy of Sciences, 1999, 96(1):290-295.
DOI URL |
| [6] |
FU Xiangdong, Duraialagaraja Sudhakar, PENG jinrong, et al. Expression of Arabidopsis GAI in transgenic rice represses multiple gibberellin responses[J]. The Plant Cell Online, 2001, 13(8):1791-1802.
DOI URL |
| [7] | Morohashi K, Minami M, Takase H, et al. Isolation and characterization of a novel GRAS gene that regulates meiosis-associated gene expression[J]. Journal of Biological Chemistry, 2003, 278(23):65-73. |
| [8] |
BOLLE C, KONCZ C, CHUA N H, et al. PAT1, a new member of the GRAS family, is involved in phytochrome a signal transduction[J]. Genes & Development, 2000, 14(10):1269-1278.
DOI URL |
| [9] |
Heo J O, Chang K S, Kim I A, et al. Funneling of gibberellin signaling by the GRAS transcription regulator SCARECROW-LIKE 3 in the Arabidopsis root[J]. Proceedings of the National Academy of Sciences, 2011, 108(5):2166-2171.
DOI URL |
| [10] |
Xue L, Cui H, Beur B, et al. Network of GRAS transcription factors involved in the Control of Arbuscule Development in Lotus japonicas[J]. Plant Physiology, 2015, 167(3):854-871.
DOI PMID |
| [11] |
Torres-Galea P, Hirtreiter B, Bolle C, et al. Two GRAS proteins, SCARECROW-LIKE21 and PHYT ochrome a signal transduction1, function cooperatively in phytochrome A signal transduction[J]. Plant Physiology, 2013, 161(1):291-304.
DOI PMID |
| [12] |
Li X Y, Qian Q, Fu Z M, et al. Control of tillering in rice[J]. Nature, 2003, 422(6932):618.
DOI URL |
| [13] |
Lee M H, Bohye K, Sang-Kee S, et al. Large-scale analysis of the GRAS gene family in Arabidopsis thaliana[J]. Plant Molecular Biology, 2008, 67(6):659-670.
DOI URL |
| [14] | Li L, Zeng W, Li Y, et al. Analysis of GRAS family transcription factors, cloning and researching cold tolerance of LaSCL18 gene in Lepidium[J]. Molecular Plant Breeding, 2017, 15(9):3428-3437. |
| [15] | Shi R, Cao Y, Chen W, et al. On cDNA cloning and expression analysis of GRAS gene in fingered citron[J]. Journal of Zhejiang Normal University, 2011, 34(4):446-451. |
| [16] | 李雪燕, 金胶胶, 赵玉琳, 等. 柽柳GRAS转录因子基因启动子克隆和表达分析[J]. 中国农学通报, 2016, 32(2):28-32. |
| LI Xueyan, JIN Jiaojiao, ZHAO Yulin, et al. Cloning and expression analysis of GRAS transcription factor gene promoter of Tamarix chinensis[J]. Bulletin of Chinese Agronomy, 2016, 32(2):28-32. | |
| [17] |
Xu K, Chen S, Li T, et al. OsGRAS23, a rice GRAS transcription factor gene, is involved in drought stress response through regulating expression of stress-responsive genes[J]. BMC Plant Biology, 2015, 15(1):141.
DOI URL |
| [18] |
Ma H S, Dan L, Peng S, et al. The salt- and drought-inducible poplar GRAS protein SCL7 confers salt and drought tolerance in Arabidopsis thaliana[J]. Journal of Experimental Botany, 2010, 61(14):4011-4019.
DOI URL |
| [19] |
Zhang S, LI X, FAN S, et al. Overexpression of HcSCL13, a Halostachys caspica GRAS transcription factor, enhances plant growth and salt stress tolerance in transgenic Arabidopsis[J]. Plant Physiology and Biochemistry, 2020, 151:243.
DOI URL |
| [20] | Zeng Y L, Cai Z Z, Ma J, et al. Effects of salt and water stress on seed germination of halophytes Kalidium foliatum and Halostachys caspica[J]. Chinese Journal of Ecology, 2006, 25(9):1014-1018. |
| [21] | Cai L, Zhang F C, Ma J, et al. Cloning and sequence analysis of NHX gene from three species of Halophytes from Xinjiang[J]. Plant Physiology Communication, 2005, 41(3):383-387. |
| [22] | Xi J B, Zhang F S, Mao D R, et al. Species diversity and distribution of halophytic vegetation in Xinjiang[J]. Scientia Siluae Sinicae, 2006, 42(10):6-12. |
| [23] | 周莲洁, 杨中敏, 张富春, 等. 新疆盐穗木GRAS转录因子基因克隆及表达分析[J]. 西北植物学报, 2013, 33(6):1091-1097. |
| ZHOU Lianjie, YANG Zhongmin, ZHANG Fuchun, et al. Cloning and expression analysis of GRAS transcription factor gene from salt spike in Xinjiang[J]. Acta Botanica Boreali-Occidentalia Sinica, 2013, 33(6):1091-1097. | |
| [24] | 樊寿德, 王艳. 盐穗木盐相关转录因子HcSCL13基因启动子的克隆及活性初步分析[J]. 生物技术通报, 2017, 33(5):131-138. |
| FAN Shoude, WANG Yan. Cloning and preliminary activity analysis of the promoter of salt related transcription factor HcSCL13 gene from Halostachys caspica[J]. Biotechnology Bulletin, 2017, 33(5):134-138. | |
| [25] |
Jefferson R A. Gus fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants[J]. The EMBO Journal, 1987, 6(13):3901-3907.
DOI URL |
| [26] | 黄伟. 番茄GRAS转录因子家族的鉴定及SlGRAS24基因的功能研究[D]. 重庆:重庆大学, 2016. |
| HUANG Wei. Identification of GRAS transcription factor family and function of S1GRAS24 gene in Tomato[D]. Chongqing: Chongqing University, 2016. | |
| [27] | 董丽莉, 赵韩生, 李利超, 等. 毛竹PeSCL3基因表达特征及其启动子活性研究[J]. 热带亚热带植物学报, 2016, 24(3):252-258. |
| DONG Lili, ZHAO Hansheng, LI Lichao, et al. Expression characteristics and promoter activity of PeSCL3 gene in Moso Bamboo[J]. Journal of Tropical and Subtropical Plants, 2016, 24(3):252-258. | |
| [28] |
Yang G Y, Gao X Q, Ma K H, et al. The walnut transcription factor JrGRAS2 contributes to high temperature stress tolerance involving in Dof transcriptional regulation and HSP protein expression[J]. BMC Plant Biology, 2018, 18(1):367-380.
DOI URL |
| [29] |
Jensen, P J. Auxin Transport Is Required for Hypocotyl Elongation in Light-Grown but Not Dark-Grown Arabidopsis[J]. Plant Physiology, 1998, 116(2):455-462.
PMID |
| [30] |
Borello U, Ceccarelli E, Giuliano G, et al. Constitutive, light-responsive and circadian clock-responsive factors compete for the different l box elements in plant light-regulated promoters[J]. Plant J, 1993, 4(4):611-619.
PMID |
| [31] | Xia Z L, Wang M P, Liu Q J, et al. Isolation and functional analysis of sulfite oxidase gene AtSO promoter from Arabidopsis thaliana[J]. Journal of Plant Physiology and Molecular Biology, 2007, 33(5):369-374. |
| [32] |
Liu B L, Sun Y, Xue J N, et al. Genome-wide characterization and expression analysis of GRAS gene family in pepper (Capsicum annuum L.)[J]. Peer J, 2018, 6:e4796.
DOI URL |
| [33] |
Yuan Y Y, Fang L C, Zhang L L, et al. Overexpression of VaPAT1, a GRAS transcription factor from Vitis amurensis confers abiotic stress tolerance inArabidopsis[J]. Plant Cell Reports, 2016, 35(3):655-666.
DOI URL |
| [34] | 陈立勇, 柴丽娟, 陈尚武, 等. 葡萄SCARECROW Like 14-Like基因的表达特征及胁迫响应研究[J]. 中国农业大学学报, 2014, 19(3):80-87. |
| CHEN Liyong, CHAI Lijuan, CHEN Shangwu, et al. Expression characteristics and stress response of SCACARECROW like 14 like gene in grape[J]. Journal of China Agricultural University, 2014, 19(3):80-87. | |
| [35] |
Dong C J, Wang Y, Yu S S, et al. Characterization of a novel rice metallothionein gene promoter:its tissue specificity and heavy metal responsiveness[J]. Journal of Integrative Plant Biology, 2010, 52(10):914-924.
DOI URL |
| [36] | 练云, 刘允军, 王国英, 等. 玉米受伤诱导基因Wip1的启动子克隆及表达分析[J]. 中国农业科学, 2014, 47(14):2889-2896. |
| LIAN Yun, LIU Yunjun, WANG Guoying, et al. Cloning and expression analysis of promoter of injury inducing gene Wip1 in Maize[J]. Chinese Agricultural Science, 2014, 47(14):2889-2896. | |
| [37] | Zhou S M, Zhang H L, Li R, et al. Function identification of the nucleotides in key cis-element of dysfunctional tapetum1 (DYT1) Promoter[J]. Frontiers in Plant Science, 2017, 8:153-160. |
| [38] |
PArk H C, Kim M L, Kang Y H, et al. Pathogen- and NaCl-induced expression of the SCaM-4 promoter is mediated in part by a GT-1 box that interacts with a GT-1-like transcription factor1[J]. Plant Physiology, 2004, 135(4):2150-2161.
DOI URL |
| [1] | 毛晓菲, 黄世平, 银芳柳, 曾幼玲. 盐穗木HcALDH7B4基因的原核表达增强耐盐抗旱性[J]. 新疆农业科学, 2023, 60(8): 2006-2012. |
| [2] | 迪拉热·海米提, 樊永红, 王伟楠, 喻文丽, 艾海白尔·卡斯木. 盐穗木叶片及根际土壤微生物群落高通量分析[J]. 新疆农业科学, 2021, 58(4): 731-740. |
| [3] | 李建平, 足木热木·吐尔逊, 常晓春, 郝晓燕, 陈果, 高升旗, 孙良斌, 黄全生. 玉米钙依赖蛋白激酶38(ZmCDPK38)的生物信息学及表达特性分析[J]. 新疆农业科学, 2021, 58(1): 49-55. |
| [4] | 艾丽曼·阿布来孜, 赵海菊, 塔伊尔·买买提江, 阿拉依·哈那提, 王长海, 兰海燕. 非生物胁迫对不同玉米杂交种苗期生长的影响[J]. 新疆农业科学, 2020, 57(1): 32-42. |
| [5] | 王鹏举, 黄世平, 杨瑞瑞, 曾幼玲. 盐胁迫下盐穗木miR393b与预测靶基因HcTIR1的表达及相关性[J]. 新疆农业科学, 2019, 56(4): 707-713. |
| [6] | 郭丁,曲延英,倪志勇,陈全家. PEG-6000和NaCl胁迫对转GbWRKY32基因烟草种子萌发及苗期生长的影响[J]. 新疆农业科学, 2018, 55(8): 1417-1427. |
| [7] | 张慧珍, 黄世平, 杨瑞瑞, 曾幼玲. 盐穗木miR167d和预测靶基因ARF8在盐胁迫下的表达与相关性分析[J]. 新疆农业科学, 2018, 55(11): 2080-2088. |
| [8] | 张冀;杜驰;张富春. 盐胁迫下盐穗木DNA聚合酶λ基因的克隆和表达分析[J]. , 2017, 54(2): 361-370. |
| [9] | 白雪芹;杨瑞瑞;曾幼玲. 不同引物对和退火温度对盐生植物盐穗木内参基因β-actin扩增效率的影响[J]. , 2017, 54(2): 352-360. |
| [10] | 王虹;齐政;张富春. 不同浓度盐胁迫下盐穗木叶片结构的比较观察[J]. , 2016, 53(11): 2098-2105. |
| [11] | 黄世平;戴玲玲;宋策;曾幼玲. 盐穗木醛脱氢酶基因HcALDH7A1原核表达载体的构建及蛋白诱导表达与优化[J]. , 2015, 52(8): 1510-1516. |
| [12] | 苏立波;艾力扎提·艾力;刘柳;彭丹;杨艺;张富春. 盐穗木Hc2a1基因的克隆及与表达分析[J]. , 2014, 51(9): 1686-1691. |
| [13] | 王丽敏;张霞;张富春. 植物激素赤霉素和萘乙酸对盐穗木种子萌发的影响[J]. , 2014, 51(3): 504-510. |
| [14] | 地丽热巴·地里夏提;张霞;庄淑珍;张富春. 盐穗木谷胱甘肽转硫酶基因的克隆和表达分析[J]. , 2014, 51(2): 318-324. |
| [15] | 张霞;张富春. 盐生植物盐穗木悬浮细胞系的建立[J]. , 2013, 50(9): 1718-1723. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||