

新疆农业科学 ›› 2023, Vol. 60 ›› Issue (6): 1451-1459.DOI: 10.6048/j.issn.1001-4330.2023.06.018
收稿日期:2022-10-28
出版日期:2023-06-20
发布日期:2023-06-20
通信作者:
单守明(1975-),男,山东临沂人,教授,博士研究生,硕士生导师,研究方向为葡萄栽培,(E-mail) fxssm@163.com
作者简介:韦伟(1996-),男,宁夏青铜峡人,研究方向为葡萄逆境生理与分子生物学,(E-mail)weiwei12312321@163.com
基金资助:
WEI Wei1,2( ), SHAN Shouming1(
), SHAN Shouming1( ), XU Wendi1, LI Guangzong1
), XU Wendi1, LI Guangzong1
Received:2022-10-28
Online:2023-06-20
Published:2023-06-20
Correspondence author:
SHAN Shouming(1975-), male, from Linyi, Shandong Province, professor, research direction: grape cultivation, (E-mail) fxssm@163.com
Supported by:摘要:
【目的】研究山葡萄生根的分子机理,为山葡萄生产繁殖中生根提供理论数据和参考依据。【方法】对山葡萄‘双优’进行组织培养至生根阶段,设置不同浓度IBA(0.1、0.3和0.5 mg/L)、NAA(0.1、0.3和0.5 mg/L)和蔗糖(15、25和35 g/L)处理,选择根系数目差别较大的两组,对其基部生根的愈伤组织进行转录组测序分析。【结果】6个样本共获得的高质量序列共284.40 Gb,89.15%~90.3%的Clean Reads 比对到参考基因组上, S6 较S1中显著上调的差异表达基因 537个,显著下调的差异表达基因 277个,共814个差异表达基因。共805个差异表达基因注释到COG的21个分类中,主要包括未知功能、细胞内运输和信号转导机制等条目。差异表达基因主要富集于多糖结合、驱动蛋白复合体和生长素激活信号通路等条目。差异表达基因富集在苯丙烷生物合成、类黄酮生物合成和植物激素信号转导等通路。生长素途径中的11个差异表达基因均为上调表达,表达量差异较大的5个基因为IAA14、IAA22、IAA27、LAX3和A-ARR基因。转录组数据可信。【结论】通过对不同浓度的IBA、NAA和蔗糖诱导‘双优’葡萄组培苗生根的愈伤组织进行测序,获得了较为完整的转录组数据,并对其进行功能注释、富集分析和激素相关基因表达量分析。
中图分类号:
韦伟, 单守明, 徐文娣, 李光宗. 山葡萄‘双优’组织培养生根期愈伤组织的转录组分析[J]. 新疆农业科学, 2023, 60(6): 1451-1459.
WEI Wei, SHAN Shouming, XU Wendi, LI Guangzong. Transcriptome analysis of callus at rooting stage in tissue culture of vitis amurensis 'shuangyou'[J]. Xinjiang Agricultural Sciences, 2023, 60(6): 1451-1459.
| 试验组 Test group | 蔗糖浓度 Sucrose concentration (g/L) | NAA浓度 NAA concentration (mg/L) | IBA浓度 IBA concentration (mg/L) |
|---|---|---|---|
| S1 | 15 | 0.1 | 0.1 |
| S2 | 15 | 0.5 | 0.3 |
| S3 | 15 | 0.3 | 0.5 |
| S4 | 25 | 0.3 | 0.1 |
| S5 | 25 | 0.1 | 0.3 |
| S6 | 25 | 0.5 | 0.5 |
| S7 | 35 | 0.5 | 0.1 |
| S8 | 35 | 0.3 | 0.3 |
| S9 | 35 | 0.1 | 0.5 |
表1 正交试验设计
Tab.1 Orthogonal experimental design table
| 试验组 Test group | 蔗糖浓度 Sucrose concentration (g/L) | NAA浓度 NAA concentration (mg/L) | IBA浓度 IBA concentration (mg/L) |
|---|---|---|---|
| S1 | 15 | 0.1 | 0.1 |
| S2 | 15 | 0.5 | 0.3 |
| S3 | 15 | 0.3 | 0.5 |
| S4 | 25 | 0.3 | 0.1 |
| S5 | 25 | 0.1 | 0.3 |
| S6 | 25 | 0.5 | 0.5 |
| S7 | 35 | 0.5 | 0.1 |
| S8 | 35 | 0.3 | 0.3 |
| S9 | 35 | 0.1 | 0.5 |
| 基因ID Gene ID | 注释 Annotated | 序列 Sequence (5'-3') |
|---|---|---|
| VIT_07s0289g00080 | 糖基转移酶 | F: GCTACACGCTTCATCTCCAAGTCC R: GAGCCGCCTTCATCATAGCCATC |
| VIT_13s0067g02360 | 过氧化物酶 | F: GGTGTGGTCTCGTGTGCTGATG R: TTGGAGGAGGGATGCTGTTGTTTG |
| VIT_18s0041g00710 | 糖基转移酶 | F: AAAGGATTGGAGAACAGCGGACAG R: ATCGGCTTCTTCACTCTGCTTGC |
| VIT_00s0615g00020 | 含PKS_ER结构域的蛋白质 | F: GAGATTGTAGGCATCGTGACAGAGG R: GCTCCAACCAGACACCCAACAC |
| VIT_14s0128g00660 | 胚蛋白样蛋白 | F: TCATGGCATTGGCTTCCTCTCTTG R: TCTTGGGTTCAGGAATGGCAACAC |
| VIT_07s0005g06450 | 含蛋白激酶结构域的蛋白质 | F: GTTTCCTTTCCGTGGCTCCAGAG R: GGCTTCCGCTACTTCTCGTTCAC |
表2 qRT-PCR引物序列
Tab.2 Primer sequences of qRT-PCR
| 基因ID Gene ID | 注释 Annotated | 序列 Sequence (5'-3') |
|---|---|---|
| VIT_07s0289g00080 | 糖基转移酶 | F: GCTACACGCTTCATCTCCAAGTCC R: GAGCCGCCTTCATCATAGCCATC |
| VIT_13s0067g02360 | 过氧化物酶 | F: GGTGTGGTCTCGTGTGCTGATG R: TTGGAGGAGGGATGCTGTTGTTTG |
| VIT_18s0041g00710 | 糖基转移酶 | F: AAAGGATTGGAGAACAGCGGACAG R: ATCGGCTTCTTCACTCTGCTTGC |
| VIT_00s0615g00020 | 含PKS_ER结构域的蛋白质 | F: GAGATTGTAGGCATCGTGACAGAGG R: GCTCCAACCAGACACCCAACAC |
| VIT_14s0128g00660 | 胚蛋白样蛋白 | F: TCATGGCATTGGCTTCCTCTCTTG R: TCTTGGGTTCAGGAATGGCAACAC |
| VIT_07s0005g06450 | 含蛋白激酶结构域的蛋白质 | F: GTTTCCTTTCCGTGGCTCCAGAG R: GGCTTCCGCTACTTCTCGTTCAC |
| 样品名称 Sample name | RNA 浓度 RNA concentration (ng/μL) | OD260/280 | OD260/230 | RIN值 RIN value |
|---|---|---|---|---|
| S1-1 | 454.50 | 2.07 | 2.19 | 10 |
| S1-2 | 387.20 | 2.10 | 1.77 | 10 |
| S1-3 | 437.10 | 2.07 | 1.87 | 10 |
| S6-1 | 199.90 | 2.15 | 1.09 | 9.9 |
| S6-2 | 316.50 | 2.09 | 1.75 | 10 |
| S6-3 | 188.90 | 2.07 | 1.78 | 10 |
表3 样品总RNA 质检
Tab.3 The sample total RNA quality test
| 样品名称 Sample name | RNA 浓度 RNA concentration (ng/μL) | OD260/280 | OD260/230 | RIN值 RIN value |
|---|---|---|---|---|
| S1-1 | 454.50 | 2.07 | 2.19 | 10 |
| S1-2 | 387.20 | 2.10 | 1.77 | 10 |
| S1-3 | 437.10 | 2.07 | 1.87 | 10 |
| S6-1 | 199.90 | 2.15 | 1.09 | 9.9 |
| S6-2 | 316.50 | 2.09 | 1.75 | 10 |
| S6-3 | 188.90 | 2.07 | 1.78 | 10 |
| 样品名称 Sample name | 原始数据 Raw reads | 过滤数据 Clean reads | 错误率 Error rate (%) | Q20 (%) | Q30 (%) | GC含量 Content (%) |
|---|---|---|---|---|---|---|
| S1-1 | 48.77 | 48. 50 | 0.02 | 98.16 | 94.36 | 46.66 |
| S1-2 | 47.93 | 47.69 | 0.02 | 98.22 | 94.5 | 46.67 |
| S1-3 | 47.61 | 47.36 | 0.02 | 98.24 | 94.56 | 46.5 |
| S6-1 | 44.48 | 44.19 | 0.03 | 98.04 | 94.07 | 46.18 |
| S6-2 | 47.89 | 47.65 | 0.02 | 98.19 | 94.38 | 45.95 |
| S6-3 | 49.24 | 49.02 | 0.02 | 98.23 | 94.47 | 46.21 |
表4 样品测序数据统计
Tab.4 Statistical of sample sequencing data
| 样品名称 Sample name | 原始数据 Raw reads | 过滤数据 Clean reads | 错误率 Error rate (%) | Q20 (%) | Q30 (%) | GC含量 Content (%) |
|---|---|---|---|---|---|---|
| S1-1 | 48.77 | 48. 50 | 0.02 | 98.16 | 94.36 | 46.66 |
| S1-2 | 47.93 | 47.69 | 0.02 | 98.22 | 94.5 | 46.67 |
| S1-3 | 47.61 | 47.36 | 0.02 | 98.24 | 94.56 | 46.5 |
| S6-1 | 44.48 | 44.19 | 0.03 | 98.04 | 94.07 | 46.18 |
| S6-2 | 47.89 | 47.65 | 0.02 | 98.19 | 94.38 | 45.95 |
| S6-3 | 49.24 | 49.02 | 0.02 | 98.23 | 94.47 | 46.21 |
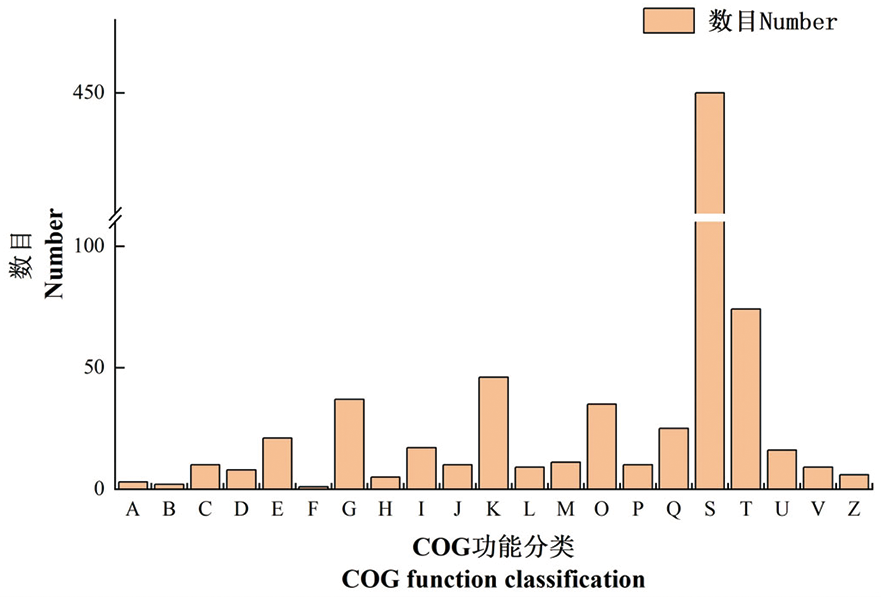
图5 差异表达基因的COG功能分类 注:A. RNA加工与修饰; B. 染色质结构与动力学; C. 能源生产和转换;D. 细胞周期控制,细胞分裂,染色体分配; E. 氨基酸转运与代谢; F. 核苷酸转运与代谢; G. 碳水化合物运输和代谢; H. 辅酶转运与代谢; I. 脂质转运与代谢; J. 翻译、核糖体结构与生物发生; K. 转录; L. 复制、重组和修复; M. 细胞壁/膜/包膜生物发生; O. 翻译后修饰,蛋白质周转,伴侣; P. 无机离子转运与代谢; Q. 次生代谢物生物合成、运输和分解代谢; S. 功能未知; T. 信号转导机制; U. 细胞内运输、分泌和囊泡转运; V. 防御机制; Z. 细胞骨架
Fig.5 COG function classification of differentially expressed genes Note: A. RNA processing and modification; B. Chromatin structure and dynamics; C. Energy production and conversion; D. Cell cycle control, cell division, chromosome partitioning; E. Amino acid transport and metabolism; F. Nucleotide transport and metabolism; G. Carbohydrate transport and metabolism; H. Coenzyme transport and metabolism; I. Lipid transport and metabolism; J. Translation, ribosomal structure and biogenesis; K. Transcription; L. Replication, recombination and repair; M. Cell wall/membrane/envelope biogenesis; O. Posttranslational modification, protein turnover, chaperones; P. Inorganic ion transport and metabolism; Q. Secondary metabolites biosynthesis, transport and catabolism; S. Function unknown; T. Signal transduction mechanisms; U. Intracellular trafficking, secretion, and vesicular transport; V. Defense mechanisms; Z. Cytoskeleton

图9 激素相关基因热图 注:使用log10(FPKM)值进行聚类分析。红色代表高表达基因,蓝色代表低表达基因,从蓝色到红色为log10(FPKM)逐渐上升
Fig.9 Heat map of genes related to hormone Note: Using log10(FPKM) value for cluster analysis, red represents high expression genes, blue represents low expression genes, log10(FPKM) gradually increases from blue to red
| [1] | 张宇, 徐智慧, 任邵琦, 等. 山葡萄F3'H基因及其启动子的克隆与表达分析[J]. 农业生物技术学报, 2021, 29(11): 2099-2108. |
| ZHANG Yu, XU Zhihui, REN Shaoqi, et al. Cloning and Expression Analysis of F3'H Gene and Promoter from Vitis amurensis[J]. Journal of Agricultural Biotechnology, 2021, 29(11): 2099-2108. | |
| [2] | Xin S, Lan D, Bao G, et al. A stress associated protein from Chinese wild Vitis amurensis, VaSAP 15, enhances the cold tolerance of transgenic grapes[J]. Scientia Horticulturae, 2021: 285. |
| [3] | 沈育杰, 赵淑兰, 杨义明, 等. 我国山葡萄种质资源研究与利用现状[J]. 特产研究, 2006,(3): 53-57. |
| SHEN Yujie, ZHAO Shulan, YANG Yiming, et al. The research and utilization on Amur grape (Vitis. amurensis Rupr) germplasm resources in China[J]. Special Wild Economic Animal and Plant Research, 2006,(3): 53-57. | |
| [4] |
Oh K E, Shin H, Lee M K, et al. Characterization and optimization of the tyrosinase inhibitory Activity of Vitis amurensis root using LC-Q-TOF-MS coupled with a bioassay and response surface methodology[J]. Molecules, 2021, 26(2): 446.
DOI URL |
| [5] | 戴彩虹, 马绍英, 李胜, 等. 山葡萄‘双红’和‘双优’的试管快繁研究[J]. 甘肃农业大学学报, 2014, 49(4): 63-68, 72. |
| DAI Caihong, MA Shaoying, LI Sheng, et al. Rapid proliferation of Vitis amurensis ‘Shuanghong’ and ‘Shuangyou’[J]. Journal of Gansu Agricultural University, 2014, 49(4): 63-68, 72. | |
| [6] | 莫银屏. 刺葡萄离体快繁体系建立及扦插生根机理研究[D]. 长沙: 湖南农业大学, 2015. |
| MO Yinping. Establishment of in vitro rapid propagation system and rooting mechanism of cutting of Vitis spinosa[D]. Changsha: Hunan Agricultural University, 2015. | |
| [7] |
崔凯, 吴伟伟, 刁其玉. 转录组测序技术的研究和应用进展[J]. 生物技术通报, 2019, 35(7): 1-9.
DOI |
|
CUI Kai, WU Weiwei, DIAO Qiyu. Application and research progress on transcriptomics[J]. Biotechnology Bulletin, 2019, 35(7): 1-9.
DOI |
|
| [8] | 石田培, 张莉. 全转录组学在畜牧业中的应用[J]. 遗传, 2019, 41(3):193-205. |
|
SHI Tianpei, ZHANG Li. Application of whole transcriptomics in animal husbandry[J]. Hereditas(Beijing), 2019, 35(7): 1-9.
DOI URL |
|
| [9] | 林茜, 高营营, 覃换玲, 等. ‘阳光玫瑰’葡萄组培脱毒快繁技术研究[J]. 果树学报, 2021, 38(3): 435-443. |
| LIN Xi, GAO Yingying, QIN Huanling, et al. Study on the technology of virus-free and rapid propagation of ‘Shine Muscat’ grape in tissue culture[J]. Journal of Fruit Science, 2021, 38(3): 435-443. | |
| [10] | 齐向丽, 师校欣, 杜国强. ‘红国王’葡萄组织培养快速繁殖[J]. 分子植物育种, 2020, 18(3): 982-987. |
| QI Xiangli, SHI Xiaoxin, DU Guoqiang. Rapid propagation of ‘Hongguowang’ grape in Vitro[J]. Molecular Plant Breeding, 2020, 18(3): 982-987. | |
| [11] | Saini S, Sharma I, Kaur N, et al. Auxin: a master regulator in plant root development[J]. Pant Cell Rep, 2013, 32(6): 741-757. |
| [12] |
Ljung K, Bhalerao RP, Sandberg G. Sites and homeostatic control of auxin biosynthesis in Arabidopsis during vegetative growth.[J]. The Plant Journal, 2001, 28(4): 465-474.
DOI URL |
| [13] |
Ikeda Y, Men S Z, Fischer U, et al. Local auxin biosynthesis modulates gradient-directed planar polarity in Arabidopsis[J]. Nature Cell Biology, 2009, 11(6):731-738.
DOI PMID |
| [14] | Robert H S, Friml J. Auxin and other signals on the move in plants[J]. Nature Chemical Ciology, 2009, 5(5): 325-332. |
| [15] | 林雨晴, 齐艳华. 生长素输出载体PIN家族研究进展[J]. 植物学报, 2021, 56(2):151-165. |
| LIN Yuqing, QI Yanhua. Advances in auxin efflux carrier PIN proteins[J]. Chinese Bulletin of Botany, 2021, 56(2): 151-165. | |
| [16] |
孙雪丽, 刘范, 田娜, 等. 香蕉Aux/IAA基因家族的全基因组鉴定及表达分析[J]. 园艺学报, 2019, 46(10): 1919-1935.
DOI |
|
SUN Xueli, LIU Fan, TIAN Na, et al. Genome-wide identification and expression analysis of Aux/IAA gene family in banana[J]. Acta Horticulturae Sinica, 2019, 46(10): 1919-1935.
DOI |
|
| [17] |
李俊男, 燕晓杰, 李枢航, 等. 植物AUX/IAA基因家族研究进展[J]. 中国农学通报, 2018, 34(15): 89-92.
DOI |
| LI Junnan, YAN Xiaojie, LI Shuhang, et al. Plants AUX/IAA gene family: research progress[J]. Chinese Agricultural Science Bulletin, 2018, 34(15): 89-92. | |
| [18] |
曾文芳, 王小贝, 潘磊, 等. 桃Aux/IAA家族基因鉴定及在果实成熟过程中的表达分析[J]. 园艺学报, 2017, 44(2): 233-244.
DOI |
|
ZENG Wenfang, WANG Xiaobei, PAN Lei, et al. Identification and expression profiling of Aux/IAA family gene during peach fruit ripening[J]. Acta Horticulturae Sinica, 2017, 44(2): 233-244.
DOI |
|
| [19] | 黎颖, 左开井, 唐克轩. 植物GH3基因家族的功能研究概况[J]. 植物学通报, 2008,(5): 507-515. |
| LI Ying, ZUO Kaijing, TANG Kexuan. A survey of functional studies of the GH3 gene family in plants[J]. Chinese Bulletin of Botany, 2008,(5): 507-515. | |
| [20] |
曾文芳, 潘磊, 牛良, 等. 桃GH3基因家族的生物信息学分析及其在果实发育中的表达[J]. 园艺学报, 2015, 42(5): 833-842.
DOI |
|
ZENG Wenfang, PAN Lei, NIU Liang, et al. Bioinformatics analysis and expression of the nectarine indole-3-aceticacid-amido synthase(GH 3)gene family during fruit development[J]. Acta Horticulturae Sinica, 2015, 42(5): 833-842.
DOI |
|
| [21] |
刘晓东, 王若仲, 焦彬彬, 等. 拟南芥IAA酰胺合成酶GH3-6负调控干旱和盐胁迫的反应[J]. 植物学报, 2016, 51(5): 586-593.
DOI |
| LIU Xiaodong, WANG Ruozhong, JIAO Binbin, et al. Indole acetic acid-amido synthetase GH3-6 negatively regulates response to drought and salt in Arabidopsis[J]. Chinese Bulletin of Botany, 2016, 51(5): 586-593. | |
| [22] | 刘帅, 徐伟荣, 张亚红, 等. 基于转录组研究补光对设施‘红地球’葡萄萌芽的影响[J]. 果树学报, 2021, 38(3): 305-317. |
| LIU Shuai, XU Weirong, ZHANG Yahong, et al. Effects of supplementary light on the bud burst of ‘Red Globe’ grape under protected cultivation based on transcriptome sequencing[J]. Journal of Fruit Science, 2021, 38(3): 305-317. | |
| [23] | 尚骁尧, 周玲芳, 石欣玥, 等. 蒺藜苜蓿细胞分裂素响应调节因子ARR9自激活检测及表达分析[J]. 中国草地学报, 2021, 43(12): 1-10. |
| SHANG Xiaorao, ZHOU Linfang, SHI Xinyue, et al. Self-activation detection and expression analysis of cytokinin response regulator ARR9 in Medicago truncatula[J]. Chinese Journal of Grassland, 2021, 43(12): 1-10. |
| [1] | 杨静, 王为然, 王萌, 朱家辉, 宁新民, 阿里甫·艾尔西, 闵玲, 孔杰. 新疆海岛棉胚性愈伤组织培养体系[J]. 新疆农业科学, 2022, 59(6): 1321-1329. |
| [2] | 杨洁,贾凯,闫会转,高杰. 芜菁离体培养再生体系建立[J]. 新疆农业科学, 2018, 55(8): 1516-1522. |
| [3] | 钟颖, 冯建荣, 樊新民 , 任欢喜, 张秀抗, 许竹叶. 库尔勒香梨离体叶片再生体系的建立[J]. 新疆农业科学, 2018, 55(5): 829-836. |
| [4] | 故丽米热·卡克什,木合塔尔·扎热,马合木提·阿不来提, 史彦江,宋锋惠. 不同浓度IBA对大果榛子不同品种黄化萌条扦插成活率影响综合评价[J]. 新疆农业科学, 2018, 55(3): 468-476. |
| [5] | 张译元, 郭延华, 王聪慧, 唐红, 南海艳, 王立民, 周平. 绵羊iPS细胞诱导及转录组学分析[J]. 新疆农业科学, 2018, 55(11): 2142-2149. |
| [6] | 伊丽米努尔;李宏. 天山云杉成熟与未成熟合子胚的胚性愈伤组织诱导及增殖研究[J]. , 2017, 54(2): 262-271. |
| [7] | 包秋娟, 张丽丽, 海那尔·乌拉孜巴依, 张富春. 干旱胁迫棉花转录组DNA损伤修复相关基因的分析[J]. 新疆农业科学, 2017, 54(11): 1999-2005. |
| [8] | 沈春修. 水稻LOC_Os10g05490位点冷胁迫条件下表达分析及CRISPR/Cas9定向编辑[J]. 新疆农业科学, 2017, 1(1): 1-10. |
| [9] | 潘越;史彦江;宋锋惠;陶秀冬. 不同因素对平欧杂种榛嫩枝扦插生根的影响[J]. , 2016, 53(6): 1034-1041. |
| [10] | 李月荣;李荣飞;张波;李迪迪;王晓琴. FeCl3溶液诱导葡萄愈伤组织白藜芦醇积累及其与氧化应激的关系[J]. , 2016, 53(1): 149-155. |
| [11] | 张燕红;赵志强;吴泽新;袁杰;布哈丽且木;王奉斌. 根癌农杆菌介导的水稻遗传转化体系的研究[J]. , 2014, 51(8): 1457-1462. |
| [12] | 张霞;张富春. 盐生植物盐穗木悬浮细胞系的建立[J]. , 2013, 50(9): 1718-1723. |
| [13] | 李鹏飞;朱华国;程文翰;王凡龙;张新宇;孙杰. 新陆早33号高效遗传转化体系研究[J]. , 2013, 50(6): 981-987. |
| [14] | 廖茂森;何金燕;张富春. 棉花新陆早17号愈伤组织中盐响应相关基因的表达分析[J]. , 2013, 50(3): 401-408. |
| [15] | 周龙;彭妮;王超;苏向辉;赵立魁;叶丹. 不同激素处理对天山樱桃绿枝扦插生根效果研究[J]. , 2013, 50(12): 2236-2240. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||